 
The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10-19 coulombs. The molar mass is used to convert grams of a substance to moles and is used often in chemistry. 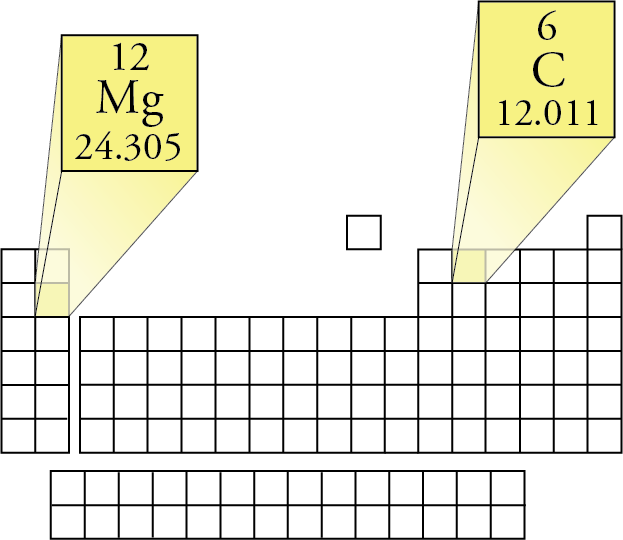
Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6.022 × 10 23 carbon atomsis therefore 12.011 g/mol: Table 2.4.1 2.4. Trending Questions How does wind break help soil? What does it mean when temperature is 35.3 on an adult? How is the luster of talc? How many sulfur dioxide molecules are there in 1. The latest release of the Periodic Table (dated ) includes the most recent abridged standard atomic weight values released by the IUPAC Commission on Isotopic Abundances and Atomic Weights (), compiled as part of the 2021 Table of Standard Atomic Weights 2021. Then, lookup atomic weights for each element in periodic table: C: 12.0107, U: 238.02891 Now, compute the sum of products of number of atoms to the atomic weight: Molar mass ((CU)) Count i Weight i Count(C) Weight(C) + Count(U) Weight(U) 1 12.0107 + 1 238.02891 250. Copper is a chemical element with atomic number 29 which means there are 29 protons in its nucleus. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
